Atoms to Moles Calculator A mole is a unit which defined as the amount of a chemical substance that contains as many representative particles. The atom is the smallest particle of a chemical element that can exist.
You cannot directly convert grams to atoms. First you must covert your grams to moles, then you can take the moles and covert to atoms. If you take your 878 grams of fluorine and then look at. Jan 13, 2008 First of all, you have to convert grams to number of moles by dividing the grams with its molar mass. After you get the molar mass, you have to understand that one mole contain 6.022x10^23 atoms.
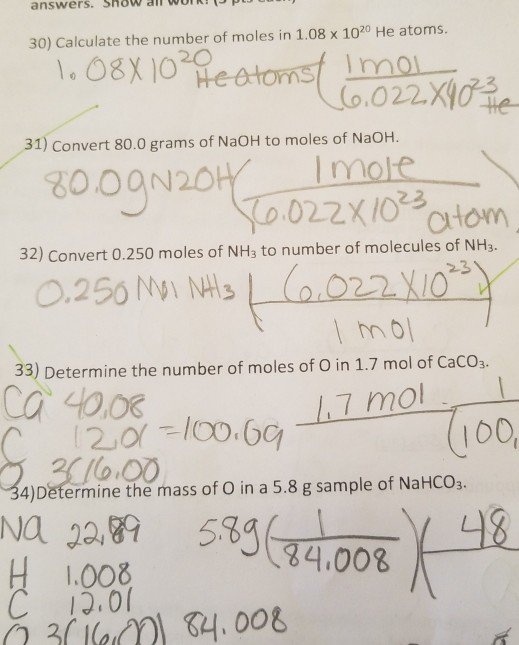
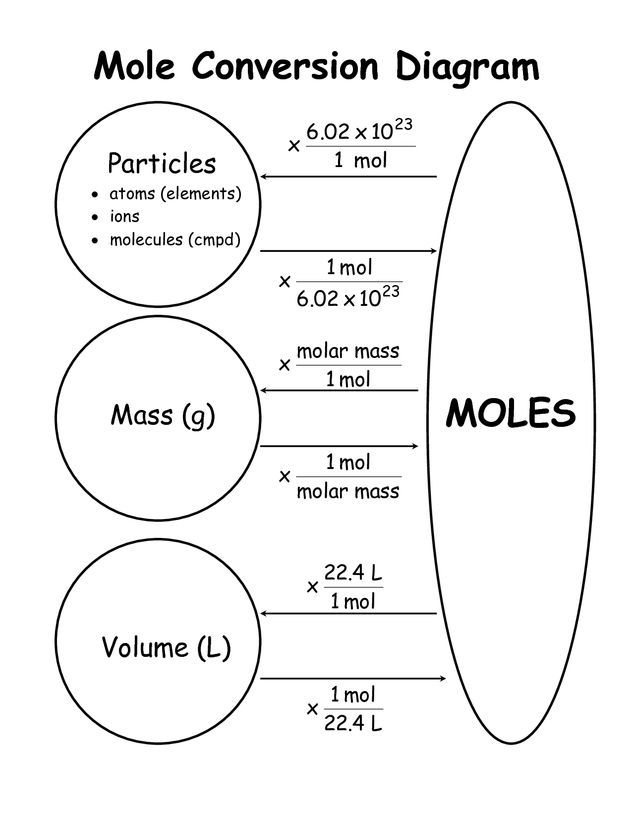
In a general chemistry class, you usually end up having to perform a lot of conversions involving moles (mol). Whether you’re converting from moles to grams, moles to volume, or moles to particles (atoms or molecules), use this quick guide to remind you of how to do each type of mole conversion:

Moles To Atoms Conversion
- Converting from mass (grams) to moles: Divide your initial mass by the molar mass of the compound as determined by the periodic table.Hp deskjet f4500 series driver. Jul 27, 2019 HP Deskjet F4500 All-in-One Printer series. Printer Support – On this page, you can find drivers, software, manual and wireless settings that are suitable for your HP product or without driver software along with an explanation of product compatibility with each operating system. Click on the individual topics below to get detailed information. Find support and troubleshooting info including software, drivers, and manuals for your HP Deskjet F4500 All-in-One Printer series. Download the latest drivers, firmware, and software for your HP Deskjet F4500 All-in-One Printer series.This is HP’s official website that will help automatically detect and download the correct drivers free of cost for your HP Computing and Printing products for Windows and Mac operating system.
- Converting from moles to mass (grams): Multiply your initial mole value by the molar mass of the compound as determined by the periodic table.
- Converting from volume (liters) to moles: Divide your initial volume by the molar volume constant, 22.4 L.
- Converting from moles to volume (liters): Multiply your mole value by the molar volume constant, 22.4L.
- Converting from particles (atoms, molecules, or formula units) to moles: Divide your particle value by Avogadro’s number, 6.02×1023. Remember to use parentheses on your calculator!
- Converting from moles to particles (atoms, molecules, or formula units): Multiply your mole value by Avogadro’s number, 6.02×1023.
- Mole-to-mole conversions: Use the coefficients from your balanced equation to determine your conversion factor. Be sure your units cancel out so you end up with the correct mole value.

